Atomic Structure

1, Plum pudding model
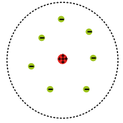
-The plum pudding model of the atom by J. J. Thomson, who discovered the electron in 1897, was proposed in 1904 before the discovery of the atomic nucleus in order to add the electron to the atomic model. In this model, the atom is composed of electrons surrounded by a soup of positive charge to balance the electrons' negative charges, like negatively charged "plums" surrounded by positively charged "pudding".
2,Rutherford model
-The Rutherford model is a model of the atom devised by Ernest Rutherford. Rutherford directed the famous Geiger-Marsden experiment in 1909 which suggested, upon Rutherford's 1911 analysis, that the so-called "plum pudding model" of J. J. Thomson of the atom was incorrect. Rutherford's new model for the atom, based on the experimental results, contained the new features of a relatively high central charge concentrated into a very small volume in comparison to the rest of the atom and with this central volume also containing the bulk of the atomic mass of the atom. This region would be named the "nucleus" of the atom in later years.
3, Bohr model
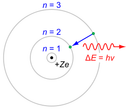
-In atomic physics, the Bohr model, introduced by Niels Bohr in 1913, depicts the atom as small, with a positively charged nucleus surrounded by electrons that travel in circular orbits around the nucleus—similar in structure to the solar system, but with attraction provided by electrostatic forces rather than gravity.
image 1:http://upload.wikimedia.org/wikipedia/commons/thumb/f/ff/Plum_pudding_atom.svg/348px-Plum_pudding_atom.svg.png
image 2:http://upload.wikimedia.org/wikipedia/commons/thumb/e/e8/Rutherford_atomic_planetary_model.svg/600px-Rutherford_atomic_planetary_model.svg.png
image 3:http://upload.wikimedia.org/wikipedia/commons/thumb/5/55/Bohr-atom-PAR.svg/310px-Bohr-atom-PAR.svg.png
text:http://en.wikipedia.org/wiki/Book:Atomic_models
image 2:http://upload.wikimedia.org/wikipedia/commons/thumb/e/e8/Rutherford_atomic_planetary_model.svg/600px-Rutherford_atomic_planetary_model.svg.png
image 3:http://upload.wikimedia.org/wikipedia/commons/thumb/5/55/Bohr-atom-PAR.svg/310px-Bohr-atom-PAR.svg.png
text:http://en.wikipedia.org/wiki/Book:Atomic_models
