Covalent Bonds
CH4
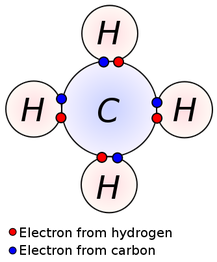
-A covalent bond is the chemical bond that involves the sharing of pairs of electrons between atoms. The stable balance of attractive and repulsive forces between atoms when they share electrons is known as covalent bonding. For many molecules, the sharing of electrons allows each atom to attain the equivalent of a full outer shell, corresponding to a stable electronic configuration.
-Covalent bonds are affected by the electronegativity of the connected atoms. Two atoms with equal electronegativity will make nonpolar covalent bonds such as H−H. An unequal relationship creates a polar covalent bond such as with H−Cl.
-Covalency is greatest between atoms of similar electronegativities. Thus, covalent bonding does not necessarily require the two atoms be of the same elements, only that they be of comparable electronegativity. Although covalent bonding entails sharing of electrons, it is not necessarily delocalized.
<- Image: http://upload.wikimedia.org/wikipedia/commons/thumb/1/17/Covalent.svg/334px-Covalent.svg.png
-Covalent bonds are affected by the electronegativity of the connected atoms. Two atoms with equal electronegativity will make nonpolar covalent bonds such as H−H. An unequal relationship creates a polar covalent bond such as with H−Cl.
-Covalency is greatest between atoms of similar electronegativities. Thus, covalent bonding does not necessarily require the two atoms be of the same elements, only that they be of comparable electronegativity. Although covalent bonding entails sharing of electrons, it is not necessarily delocalized.
<- Image: http://upload.wikimedia.org/wikipedia/commons/thumb/1/17/Covalent.svg/334px-Covalent.svg.png
